 
Subsequent reaction with pyridine and heat forms the α, β‑unsaturated ketone, 2-methyl-2-cyclopentenone. Although the enol intermediate causes a racemic mixture of the α-brominated compound to form, it is irrelevant because the chiral carbon is subsequently converted to an achiral alkene. Because enol tautomers prefer to form on the more substituted α-carbon, α-bromination also occurs on the more substituted α-carbon. In order to promote an E2 reaction, a sterically hindered base, pyridine, is often used.Īn example of this reaction involves the α-bromination of 2-methylcyclopentanone to form 2-bromo-2-methylcyclopentanone. This reaction takes place by an E2 elimination mechanism and creates a C=C double bond which is conjugated with the carbonyl. Only in the presence of an acid catalyst and heat can this reaction take place. Skill 12.3 Compare the stability of different carboxylic acid derivatives to determine reactivity. When a carboxylic acid reacts with an alcohol, esterification occurs. Skill 12.2 Draw the structure of carboxylic acid derivatives from the IUPAC name. Skill 12.1 Name carboxyilic acid derivatives using IUPAC rules. The product of an α-bromination can be converted to an α, β‑unsaturated carbonyl by reaction with pyridine and heat which causes the elimination of H and Br. Mass spectra of carboxylic acid derivatives show a base peak due to the acylium ion. These solutions for Aldehydes, Ketones And Carboxylic Acids are extremely popular among class 12 Science students for Chemistry Aldehydes, Ketones And Carboxylic Acids Solutions come handy for quickly completing your homework and preparing for exams. Although slower reacting than acid chlorides, anhydrides react with water to form carboxylic acids, with alcohols to form esters, and with amines to form amides. 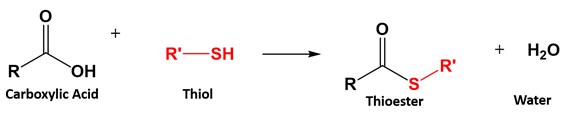
Synthetic Uses for α-Halogenated Carbonyls Anhydrides are highly reactive to nucleophilic attack and undergo many of the same reactions as acid chlorides that were explored in section 21.4. The fact that the substitution occurs on the α-carbon led Lapworth to propose that the more reactive form was an enol tautomer of acetone. These observations led Lapworth to theorize that the rate-determining step of the mechanism involves converting acetone to a more reactive form. B(OCH2CF3)3, prepared from readily available B2O3 and 2,2,2-trifluoroethanol, is as an effective reagent for the direct amidation of a variety of carboxylic acids with a broad range of amines. This implies that the halogen participates in the mechanism through a fast step which occurs after the rate-determining step. The rate law expression for the α-halogenation of a ketone can be given by: Also, it was shown that the rates for all three halogenation reactions were first-order with respect to acetone and the acid catalyst but independent of the halogen concentration (overall second-order for the mechanism). In the early 1900's chemist Arthur Lapworth showed that the rates of chlorination, bromination, and iodination of acetone were all the same. This reaction was the focus of one of the first mechanistic investigations in organic chemistry. \)Įxperimental Evidence of the Enol Intermediate 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
